|
“… I found myself looking at the table in almost geographic terms, as a realm, a kingdom, with different territories and boundaries. With samples of each element arranged in a display that followed the organization of the Periodic Table, he later described his reaction thusly: Famed author and neurologist Oliver Sacks noted this separation almost immediately when, as a boy, he visited a wall-sized exhibit of samples of all of the elements at the Science Museum near his home in London. 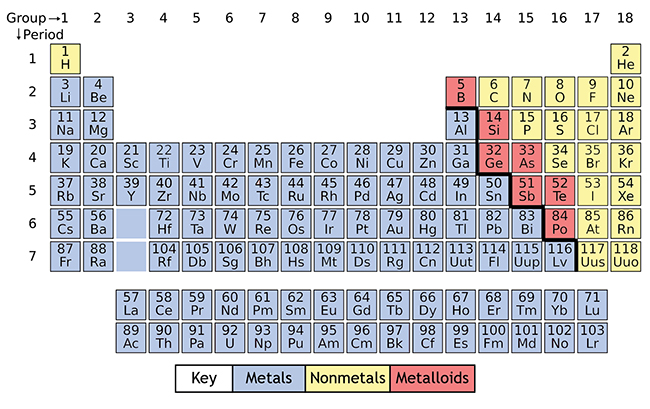
Moreover, the metallic elements seem to be spatially segregated from other elements, as illustrated by the color-coding of Figure 4-2. A cursory look reveals one thing immediately: most of the elements are metallic. Understanding some simple rules for how individual atoms interact is enormously helpful when trying to make sense of all that richness.Īn intuitively accessible approach to begin understanding the information in the Periodic Table begins with an examination of the physical characteristics of the elements. It is beyond dispute that compounds are vastly richer in their properties than elements: tens of millions of unique compounds have been identified, and the list is growing by hundreds every day, while the number of naturally occurring elements is only ninety. But, to counter his analogy a bit, one still needs to understand how letters can combine to make words: rules exist and your efforts as a reader and writer are made much easier if you can recognize patterns that provide clues as to what they are. His point is compelling and borne out by the experience of most chemists: we spend far more time thinking about what molecules do (or don’t do, if they stubbornly resist acting how we think they should) than about isolated atoms or pure elements. In other words, knowing the elements is fine and dandy, but chemistry is a rich field only because of the richness of the compounds the elements make. Likewise, molecules have far more variety and can do far more interesting things than atoms. Despite the fundamental importance of letters in any written language, things don’t get interesting until one focuses on words. As part of an argument against the traditional emphasis on the Periodic Table – sacrilege to many in our trade! - he likens atoms to letters and molecules to words. Philip Ball, in his book Stories of the Invisible, discusses how the structure of chemistry parallels that of languages. For clarity, elements 57-70, and 89-118 are omitted from this version of the Periodic Table. 
See Note 1 for comments on this labeling scheme of the vertical columns. Elements here are color coded: metallic elements are blue, nonmetals are pink, and metalloids are purple. Also note the highlighted column labels elements in columns 1A-8A are referred to as Main Group Elements, those in 1B-8B are referred to as Transition Metals. \)įigure 4-2: The "twin-towered" edifice we know as the Periodic Table of the Elements embeds a wealth of information.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
